Stage 4 Pancreatic Cancer Survival Rate, Treatment Options, and the Science Behind Daraxonrasib (2026)
Part of the Ben Sasse Pancreatic Cancer Pillar Series | Updated April 10, 2026
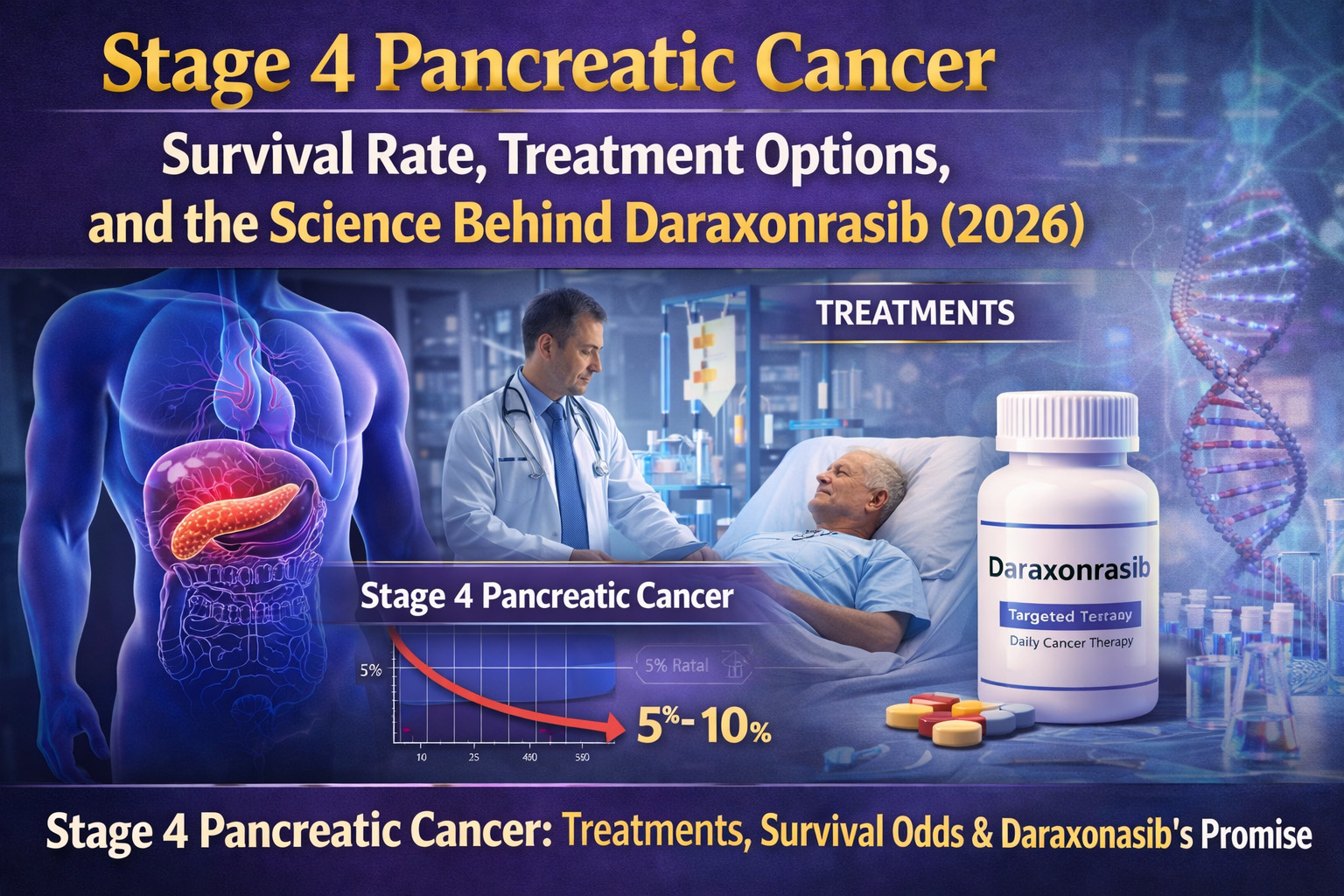
Stage 4 pancreatic cancer has a 5-year survival rate of approximately 3–5%, making it one of oncology’s most difficult challenges. This page explains why the prognosis is so poor, what treatment options exist in 2026, and why the drug daraxonrasib, the same experimental therapy former Senator Ben Sasse is taking, represents one of the most promising new approaches in the field.
Why Is Stage 4 Pancreatic Cancer So Hard to Treat?
Pancreatic cancer doesn’t play fair. Unlike some cancers that declare themselves early through visible symptoms, pancreatic tumors often grow silently for years. The pancreas sits tucked behind the stomach, deep in the abdomen, out of reach of most routine screening tools. The symptoms it eventually produces (back pain, subtle digestive issues, mild fatigue) mimic dozens of more common conditions.
By the time a diagnosis is confirmed, roughly 50-55% of patients already have stage 4 disease, meaning the cancer has spread beyond the pancreas to distant organs like the liver, lungs, or peritoneum. At that point, curative surgery isn’t possible. There are simply too many sites of disease to address surgically.
Making matters worse, pancreatic tumors create what oncologists call a “desmoplastic stroma,” a dense, fibrous barrier around the tumor that blocks most chemotherapy drugs from even reaching the cancer cells. The tumor essentially builds its own wall. As Ben Sasse’s oncologists have described it to him, fighting pancreatic cancer is like chipping at a giant dam with a pickax. Progress is real but agonizingly slow.
Stage 4 Pancreatic Cancer Survival Rate: The Real Numbers
The statistics are sobering. According to data from the National Cancer Institute’s SEER database, cited by both NCCN and ASCO:
- 5-year survival rate for stage IV pancreatic cancer: approximately 3–5%
- Median survival after a stage IV diagnosis: 6–12 months without treatment
- With modern chemotherapy (FOLFIRINOX or gemcitabine/nab-paclitaxel): median survival extends to 8–11 months
- Estimated new U.S. pancreatic cancer diagnoses in 2025: 67,440 (American Cancer Society)
- Estimated U.S. deaths from pancreatic cancer in 2025: approximately 51,980
Those numbers explain why Sasse described his diagnosis with the phrase he used. They also explain why any treatment that meaningfully extends survival, like daraxonrasib, appears to be doing for Sasse, generates genuine excitement in the oncology community.
To be clear about context: pancreatic cancer accounts for about 3% of all new U.S. cancer cases but is responsible for roughly 7% of all cancer deaths. It’s projected to overtake colorectal cancer as the second leading cause of cancer death in the U.S. by 2030.
What Is Daraxonrasib? The Drug at the Center of Sasse’s Clinical Trial
Daraxonrasib is an experimental targeted therapy developed by Revolution Medicines, a clinical-stage biopharmaceutical company based in Redwood City, California. It’s part of a newer class of cancer treatments called RAS inhibitor drugs that target a protein called RAS, which is mutated in the majority of pancreatic cancers.
Here’s why that matters: KRAS mutations are found in approximately 90% of pancreatic cancers, and for decades, the KRAS protein was considered “undruggable” its surface was too smooth for drugs to bind to effectively. The development of RAS inhibitors like daraxonrasib represents a genuine breakthrough in addressing that long-standing challenge.
Daraxonrasib is taken orally, a practical advantage for patients like Sasse who don’t want to spend their remaining months in a hospital. But the side effects are significant. The drug disrupts the skin’s ability to regenerate, causing continuous bleeding from the face and other areas. Sasse has been photographed and appeared in interviews with visible dried blood on his face, which he’s addressed with characteristic matter-of-factness.
As of April 2026, daraxonrasib has reduced Ben Sasse’s total tumor volume by 76% from late December 2025, a dramatic response for a disease with historically poor treatment outcomes.
It’s important to note what this means and what it doesn’t: a 76% reduction in tumor volume is a remarkable response, but it doesn’t mean the cancer is gone or cured. Sasse himself has used a vivid metaphor to explain the situation it’s like clearing the dandelions in a yard that has already seeded the neighboring yards. You can eliminate what’s visible, but the spread has already happened.
His oncologists have been honest: this doesn’t end well. But it may end later than originally feared — and the data generated from Sasse’s treatment, and others in the same trial, will inform the next generation of therapy.
Current Standard-of-Care for Stage 4 Pancreatic Cancer
Outside of clinical trials, the standard first-line treatment options for stage 4 pancreatic cancer in 2026 include:
FOLFIRINOX
A combination chemotherapy regimen of fluorouracil, leucovorin, irinotecan, and oxaliplatin. It’s considered one of the more aggressive standard approaches and has shown median survival benefit of approximately 11 months in clinical trials. However, it’s only suitable for patients with good baseline health (“good performance status” in oncology terminology) because the side effects are significant.
Gemcitabine + Nab-Paclitaxel
The other main first-line option, often used for patients who can’t tolerate FOLFIRINOX. It has shown comparable but slightly lower efficacy and is generally better tolerated.
Clinical Trials
For patients who have good performance status and access to major cancer centers like MD Anderson, Memorial Sloan Kettering, or Johns Hopkins, clinical trials represent a genuine option and sometimes the best one. Sasse’s experience with daraxonrasib illustrates both the potential and the difficulty: remarkable short-term tumor response, serious side effects, and no guarantee of long-term survival.
Emerging Research: What’s on the Horizon?
Pancreatic cancer research has accelerated meaningfully in the past few years:
NKT Cell Immunotherapy (UCLA)
Researchers at UCLA recently reported encouraging results from an experimental cell-based immunotherapy using natural killer T (NKT) cells, genetically modified to recognize and attack pancreatic cancer cells. In mouse studies, the therapy slowed tumor growth, extended survival, and remained effective even after the cancer had spread to the liver and lungs. Human studies are pending.
Liquid Biopsy for Early Detection
Several research groups are working on blood tests capable of detecting pancreatic cancer at earlier stages before symptoms appear and before metastasis occurs. A reliable liquid biopsy for pancreatic cancer would be transformative; it’s the detection problem, more than the treatment problem, that drives most deaths from this disease.
Combination KRAS Inhibitor Strategies
Revolution Medicines and other companies are investigating combination approaches using KRAS inhibitors alongside immunotherapy, with the hypothesis that suppressing tumor growth while simultaneously boosting immune response could produce more durable outcomes.
Psychological and Palliative Dimensions: What Ben Sasse’s Story Teaches Us
There’s a medical story here, and there’s a human story. They’re not separate.
Ben Sasse has been remarkably candid about the psychological experience of a terminal diagnosis the “heaviness” he described about not seeing his children grow up, the redirected priorities, the strange peace that has accompanied the clarity. In multiple interviews, he’s described how the diagnosis has sharpened his sense of what matters and diminished his interest in what doesn’t.
Palliative care, managing symptoms, maintaining quality of life, supporting patients and families through terminal illness, is increasingly recognized as a critical component of cancer care. It’s not “giving up.” Sasse takes morphine. He sleeps 15-16 hours a day. He chooses carefully which hours he spends and with whom.
His podcast, his interviews, his WSJ columns these aren’t denial. They’re a philosophy: that the process of dying is still something to be lived.
Key Questions This Page Answers
What is the survival rate for stage 4 pancreatic cancer?
The 5-year survival rate for stage 4 (metastatic) pancreatic cancer is approximately 3–5%, according to NCCN, ASCO, and SEER data. Median survival after diagnosis is typically 6–12 months, extendable to 8–11 months with aggressive first-line chemotherapy.
How does daraxonrasib work?
Daraxonrasib is a targeted RAS inhibitor that blocks the activity of mutated KRAS protein, which drives tumor growth in approximately 90% of pancreatic cancers. It’s taken orally and is currently in clinical trials at centers including MD Anderson Cancer Center in Houston.
Can stage 4 pancreatic cancer go into remission?
Complete remission at stage 4 is extremely rare. The goal of treatment at this stage is typically to extend survival, manage symptoms, and maintain quality of life not cure. Partial responses (like the 76% tumor volume reduction in Ben Sasse’s case) are notable but do not represent remission in the clinical sense.
What are the early warning signs of pancreatic cancer?
Early pancreatic cancer often produces no symptoms. When symptoms do appear, they can include: persistent back or abdominal pain (Sasse’s initial symptom), unexplained weight loss, jaundice (yellowing of skin/eyes), new-onset diabetes, loss of appetite, and digestive changes. Anyone experiencing a combination of these symptoms, particularly back pain combined with weight loss, should discuss pancreatic evaluation with their doctor.
Further Reading and Authoritative Resources
- National Cancer Institute: Pancreatic Cancer Treatment (PDQ)
- MD Anderson Cancer Center: Pancreatic Cancer
- American Cancer Society: Pancreatic Cancer
- Fox News Health: Pancreatic Cancer in the Spotlight After Former Senator’s Diagnosis
- Revolution Medicines: Daraxonrasib Pipeline
- Pancreatic Cancer Action Network (PanCAN)
Return to the pillar page – “Ben Sasse Pancreatic Cancer Battle Explained” – for the full story of his diagnosis, public reaction, and health updates through April 2026.